After years of dramatic twists and turns, the IVDR date of application has come and gone rather unceremoniously on Thursday, 26th May 2022.
In our previous post on the IVD series, we wanted to shed light to the transitional provisions of IVDR article 110(3). We discussed under what conditions may IVDD compliant (‘legacy’) devices still be placed on the market. We would like to update you to the latest information in light of recent MDCG guidance, as well as discuss what is going in Switzerland.
The first step: To benefit from the transitional provision, you need a valid declaration of conformity to IVDD drawn up prior to the IVDR date of application. Also, you need to have classified the device according to the IVDR Annex VIII rules to determine which transitional period applies using IVDR article 110, and as summarized here.
Devices with valid IVDD EC certificate issued by Notified Body
Devices with valid EC certificates issued by a Notified Body under the In Vitro Diagnostic Medical Devices Directive, IVDD 98/79/EC can be placed on the market or made available on the market until their certificates expire, but not longer than:

Now once you have established the transitional period, be aware that some of the IVDR requirements do apply to your legacy device according to article 110(3). Those are the requirements relating to post-market surveillance, market surveillance, vigilance, registration of economic operators and of devices. Also, make sure to evaluate any planned changes in design and intended use. Any significant change will void the legacy status and require a recertification under IVDR.
Around the date of application, we saw a surge of publications of IVD specific guidance documents, including those two crucial puzzle pieces to help manage the transitional phase without regulatory hick-ups:
“MDCG 2022-8 Regulation (EU) 2017/746 – application of IVDR requirements to ‘legacy devices’ and to devices placed on the market prior to 26 May 2022 in accordance with Directive 98/79/EC”
This guidance does a good job in explaining how to deal with the applicable IVDR requirements for legacy devices. Our hot picks from this guidance include:
- A PMS plan and report according to article 80 is required for all legacy devices, regardless of classification – unless manufacturer voluntarily prepares a periodic safety update report (PSUR) pursuant to Article 81 for class C and D devices
- Summary of safety and performance per article 29 is not required for legacy devices
- Requirements for economic operators apply only partially – those related to PMS, vigilance, and registrations.
“MDCG 2022-6 Guidance on significant changes regarding the transitional provision under Article 110(3) of the IVDR”
This guidance provides decision trees to determine which changes are considered “significant” and thus require a recertification under IVDR. Here are some examples of significant changes according to the guidance:
- Changes that alter the device’s operating principle (e.g., change from immunofluorescence to ELISA)
- New or major change of operating system or any software component
- Change of material which is essential to the operating principle
- Addition of specimen type(s) to the intended purpose
There we have it. Management of legacy devices according to IVDR article 110(3) – the confinis recipe:
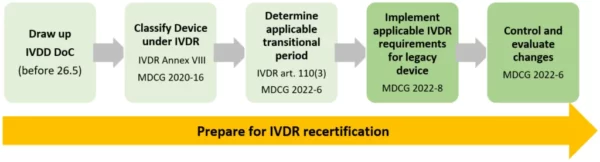
And for the time beyond transition – time to start closing the gaps and to contact an IVDR certified notified body as soon as possible – a bottleneck in IVDR certifications is heading our way at the end of the transition periods.
What about Switzerland?
The IVDR date of application and the lack of mutual recognition agreement between Switzerland and the EU, sealed Switzerland’s position as a third country to the EU IVD market. A new Swiss law (SR 812.219) for IVDs has entered into force.
The CE mark remains the accepted and required regulatory certification for selling IVDs in Switzerland. The transitional periods for legacy devices are also integrated into the Swiss law. However, additional steps are now required for both IVDD and IVDR devices:
- Mandating a Swiss Authorized Representative (“CH-REP”) to assume defined regulatory responsibilities on the Swiss market
- Labeling requirements to identify the CH-REP and the Swiss importer
- Registration of economic operators and eventually devices with Swissmedic
Transition periods for the appointment of a CH-REP, for labeling and registrations are defined as follows, according to the classification of the device by the IVDR rules:


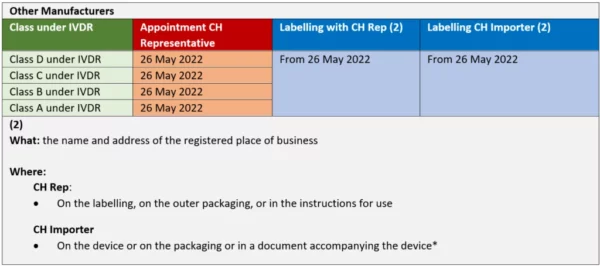
*NOTE: A “document accompanying the device” can be affixed to the device or be separate from the device. Examples of documents accompanying the device include delivery note, guarantee certificate, customs documents, invoice, a sticker on the packaging or the instructions for use. Such documents must accompany the devices through the supply chain to the distributors, however, not necessarily need to reach the end user.
Deadlines for Registration
The CH manufacturers, CH representatives and CH importers must register with Swissmedic and obtain a CHRN Swiss registration number according to the following timelines:

CH distributors do not need to register.
confinis’ services – CH-REP, PRRC and much more
For medical devices, the Swiss industry and regulators have been adapting to the new regulatory environment for a year, and it has not been without difficulty. Although the Swiss law has been written to closely imitate the IVDR, we have already seen differences in interpretation of some requirements by the regulatory bodies, for example on the central definition of “placing on the market”.
Keeping track of the deadlines, requirements and subtle legal and interpretational differences is a challenge. At confinis we are proud to combine the experience gained as CH-REP for medical devices, and the expertise of our IVD specialists to offer you a hands-on, effective, and efficient partner for your IVD CH-REP, PRRC and IVDR implementation needs. Don’t hesitate to contact us!
You can also meet us and learn more about Economic Operator Requirements under IVDR in the EU, Switzerland, and the UK at the MedTech Summit IVD Stream, 23-24.6 in Dublin.
Please contact emilia.berg@confinis.com to schedule a personal meeting.
Author: Emilia Berg, Senior Consultant

