AI is now firmly inside medicine development, but regulators are making one thing very clear:
AI is a tool, not a shortcut.
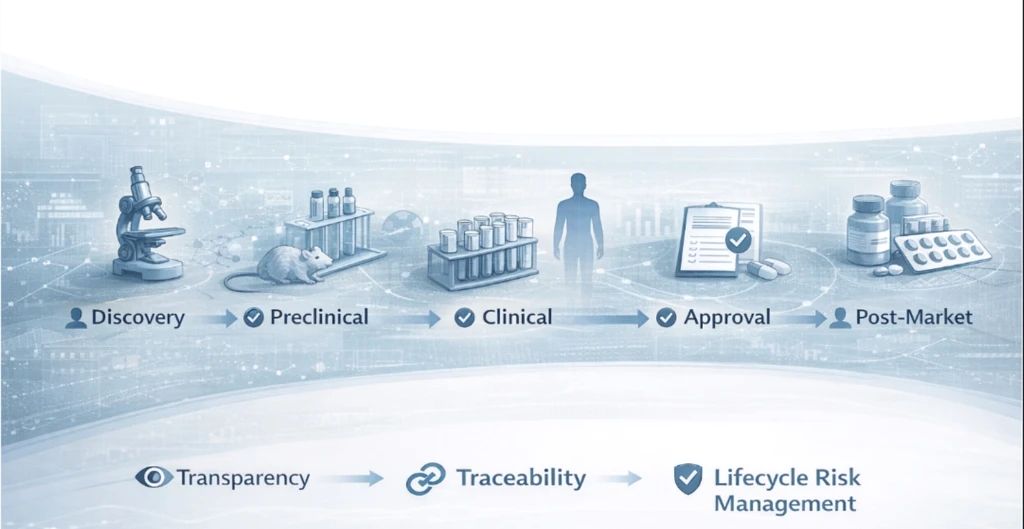
Recent communications from EMA and FDA point to a converging perspective across the lifecycle: AI can support evidence generation, analysis, and efficiency, but scientific accountability stays human.
What regulators are consistently emphasizing:
🟥 transparency of AI models
🟥 traceability of data, assumptions, and outputs
🟥 human oversight at critical decision points
🟥 lifecycle risk management, not one-off validation
This resets expectations.
The future is not “AI-driven approval.”
It’s AI-supported evidence, inspectable, explainable, and defensible under scrutiny.
Teams that treat AI as an evidence amplifier, not an evidence substitute, will move faster, and safer.
Where are you already using AI in development, and where do you expect regulators to look most closely next?
While you focus on innovation, we take care of the regulatory path!