28/04/2026 -
EUDAMED Devices Registration
24/03/2026 -
Regulatory Pathway for Combination Products
18/03/2026 -
AI Under Joint Regulatory Scrutiny
11/03/2026 -
The Swissdamed registration deadline is approaching
10/03/2026 -
Drug-device combinations regulatory pathway
05/03/2026 -
Insulin delivery as an integrated ecosystem
03/03/2026 -
Regulatory rescue for manufacturers
27/02/2026 -
AI doesn’t just hallucinate… it agrees.
25/02/2026 -
Medical devices regulatory guidance with confinis
19/02/2026 -
Can AI write Technical Documentation?
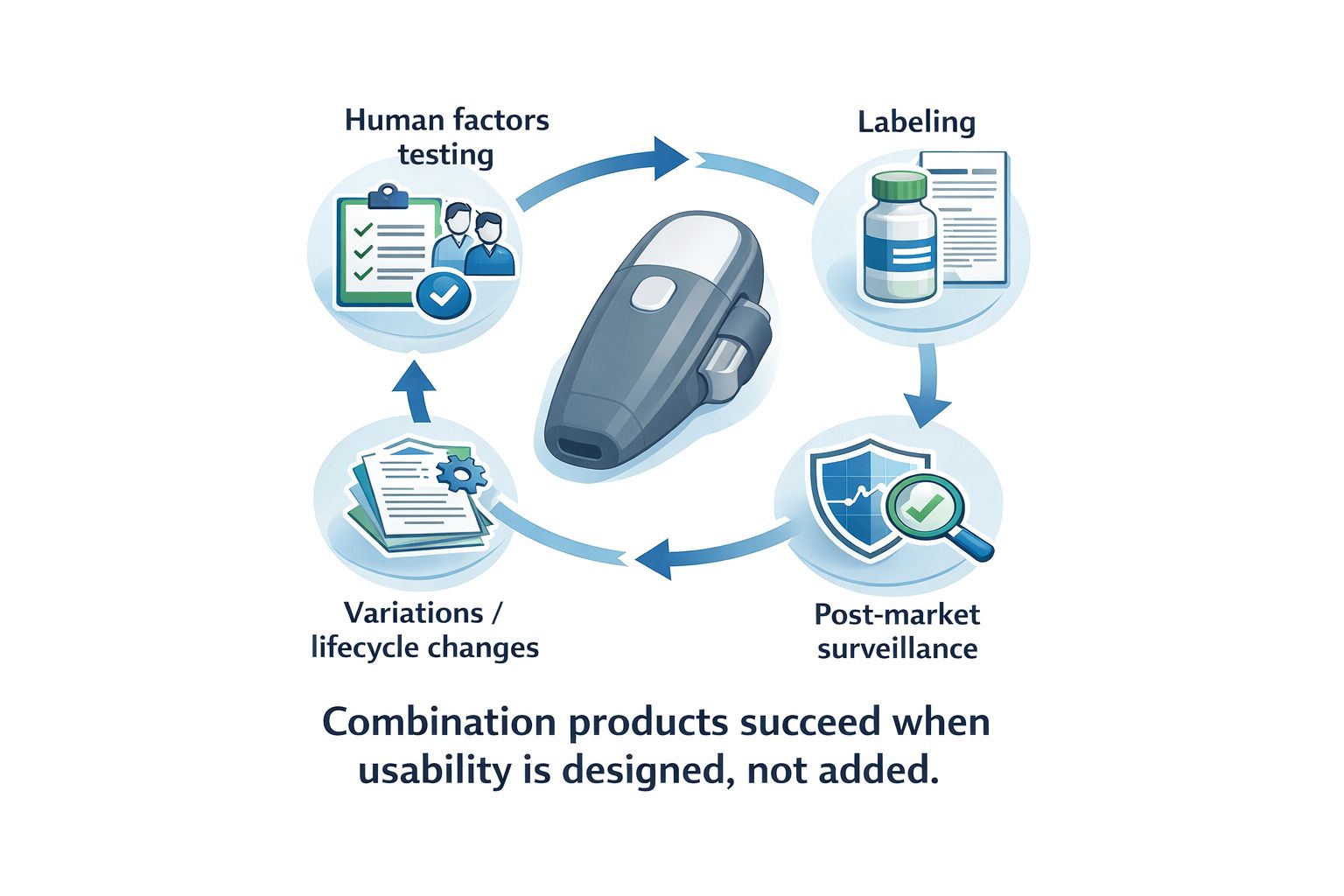
10/02/2026 -
Usability for Combination Products
30/01/2026 -
AI and Medicine Development
27/01/2026 -
AI and Regulatory Conformity
21/01/2026 -
Why did adaptive DBS scale so well in Europe?
06/10/2025 -
White Paper on Audits from a Lead Auditor’s Perspective
21/08/2025 -
White Paper on sustainability in the medical device industry
10/07/2025 -
RAPS Global Board Director Nomination Announcement
01/07/2025 -
White Paper on MDCG 2019-11 REV.1 update
15/04/2025 -
FDA Regulatory Pathways online course
30/01/2025 -
White Paper on ISO 42001:2023 and AI-Driven Medical Devices
17/01/2025 -
White Paper on FDA’s Draft Guidance for AI in Healthcare
13/01/2025 -
White Paper on Post-Market Surveillance and Vigilance
09/12/2024 -
Aligning ISO 13485 with MDR
05/11/2024 -
Regulatory Challenges for AI-Enabled Medical Devices
31/10/2024 -
White paper on Artificial Intelligence and Auto-Injectors
15/10/2024 -
QMSR and FDA QSR & ISO 13485 Harmonization
07/10/2024 -
Regulatory Compliance for AI-Enabled Medical Devices
23/09/2024 -
Medical Devices Cybersecurity Checklist
06/08/2024 -
Medical Devices Threat Modeling
23/07/2024 -
New confinis Cybersecurity Services
24/05/2024 -
(EU) 2023/607, 26th May 2024 deadline approaching
23/05/2024 -
The Future of Health Grant welcomes confinis as a new Program Partner
01/05/2024 -
Transitioning to the Medical Device Regulation (MDR) 2017/745
11/04/2024 -
Deadline for transitioning to the Medical Device Regulation
13/03/2024 -
A Step Forward in Medical Device Manufacturing: the New QMSR Aligned with ISO 13485:2016
08/03/2024 -
The EU Artificial Intelligence Act and Medical Device Regulation with Andrea Biasiucci
21/02/2024 -
Leveraging Real-World Evidence: Insights and Guidance for Medical Device Manufacturers
07/09/2023 -
confinis ag appoints Dario Motti as lead of new Digital Health Competence Cluster
03/07/2023 -
Change in Management Structure with Appointment of New CEO at confinis ag
02/12/2022 -
The Medical Device Regulation and the medical care for children and rare diseases
09/11/2022 -
Working at confinis and the IVDR world with Barbara Jeroncic
07/07/2022 -
IVD News – IVDR Date of Application, new Guidance Documents
15/06/2022 -
confinis and deep tech with Andrea Biasiucci
10/06/2022 -
Electromagnetic Compatibility (EMC) of Medical Devices
22/02/2022 -
IVD News – Progressive Roll-Out and MDCG 2022-2 on Clinical Evidence
20/12/2021 -
UK Responsible Person Services
11/10/2021 -
confinis launches new UK Responsible Person service in UK
08/10/2021 -
ISO/FDIS 11608 series opened for voting
06/07/2021 -
confinis partners with Irdeto to launch the first cybersecurity assessment tool in the medical devices industry
18/06/2021 -
confinis offers Swiss Representative Service
26/05/2021 -
Exporting Swiss Medical Devices to the EU
24/04/2021 -
Join our Startup Workshop 2021!
23/04/2021 -
… and Report your Post-Market Clinical Follow-up Evaluation!
10/03/2021 -
How to plan your post-market clinical follow up…
18/02/2021 -
How does the notified body assess your clinical evaluation?
28/01/2021 -
How to perform a clinical evaluation for Medical Device Software
15/01/2021 -
How to ensure sufficient clinical evidence for legacy devices
21/12/2020 -
How to claim an equivalence in the clinical evaluation under the MDR
10/12/2020 -
Novelties for Clinical Evaluation and Post Market Surveillance within MDR – the Posts Series
03/12/2020 -
Bei Medizinprodukten auf die EU warten hilft der Schweiz wenig
29/10/2020 -
Clinical and Performance Evaluation – our 5 strengths at your service
16/09/2020 -
confinis partners with b-rayZ concerning innovative Artificial Intelligence project
02/09/2020 -
Join us at world’s leading AI in medicine summit!
14/08/2020 -
Revision der Schweizer Medizinprodukteverordnung mit Folgen
13/07/2020 -
Workshop: Software as a Medical Device (SaMD) in Zürich
02/06/2020 -
Interpellation Jacqueline de Quattro on medical devices
02/06/2020 -
confinis celebrates 15th anniversary
13/05/2020 -
Web based tool to analyze effect of significant changes to devices covered by MDD or AIMDD certificates
08/05/2020 -
Politics intervene regarding medical devices
07/05/2020 -
Development of medical devices to tackle COVID-19 – UPDATE
01/05/2020 -
Swiss Medtech Umfrage: Export in die EU mit Drittstaatanforderungen sichern
27/04/2020 -
Peter Studer joins confinis as Senior Advisor
14/02/2019 -
EU MDR audit requirements and links to MDSAP program
27/12/2018 -
From “Essential Requirements” to “General Safety and Performance Requirements” – EU MDR 2017/745
20/12/2018 -
Technical Documentation for medical devices under the new European MDR
03/12/2018 -
Conformity Assessment Procedures and Premarket Scrutiny under EU MDR
12/11/2018 -
Classification of medical devices under the European Medical Device Regulation 2017/745
22/10/2018 -
#MDR – Transition periods and definitions
22/09/2018 -
Work in progress: MDR implementation – impact on your Quality Management System (ISO 13485:2016)
04/09/2018 -
Your insight to: confinis and its partners – work in progress regarding MDR implementation
03/09/2018 -
MDR Article 117 – a look into the Crystal Ball (Part 6)
03/09/2018 -
From Investigator to Consulting: On the other side of the fence
10/07/2018 -
MDR Article 117 – a look into the Crystal Ball (Part 5)
29/06/2018 -
WORKSHOP: PMS under the new European MDR
26/06/2018 -
“Quick Wins” for your MDR implementation
25/06/2018 -
MDR Article 117 – a look into the Crystal Ball (Part 4)
19/06/2018 -
MDR Article 117 – a look into the Crystal Ball (Part 3)
11/06/2018 -
MDR Article 117 – a look into the Crystal Ball (Part 2)
08/06/2018 -
MDR Article 117 – a look into the Crystal Ball (Part 1)
03/04/2018 -
confinis opens US office in the Washington DC area
06/02/2018 -
MDR Article 117 – are Combination Product Manufacturers prepared?
10/08/2017 -
Beat Steffen to speak at Medtech & Pharma Platform in Basel
10/08/2017 -
Upcoming ISO Technical Report regarding Post Market Surveillance
15/02/2017 -
Medical Device Regulation (MDR) and In Vitro Diagnostic Device Regulation (IVDR): Impact on Switzerland